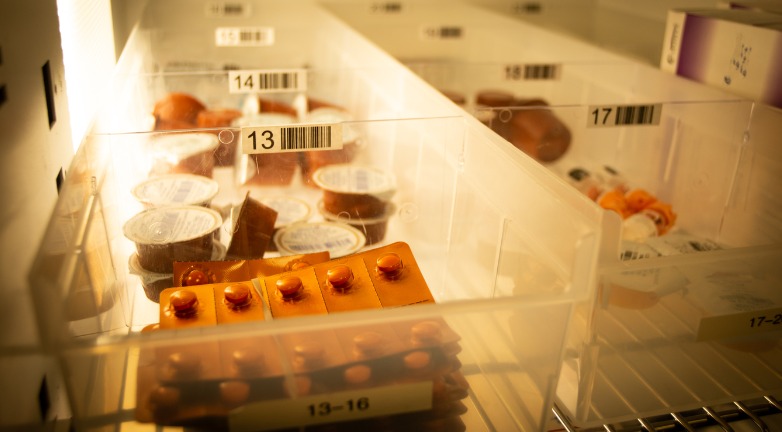
Maintaining temperature integrity and accurate product documentation can sometimes be a challenge for the logistics industry as products change hands and storage locations multiple times. However, these are important considerations that, in some cases, could have a significant impact on the end consumer’s health.
Zyma Islam writes for The Daily Star, a Bangladesh-based publication, about some cold chain issues in the country’s pharmaceutical supply chain. About a year ago, an investigative police unit discovered that two distributors, LifeTech and VieTech Lab, were selling drugs and diagnostic reagents that had not been stored at the proper temperatures. This could cause the products to denature and not function as intended. In addition, the companies did not have any records of where and when the products were manufactured, which made it harder to determine which products were expired.
The investigators then examined the sales records for the two companies to identify affected hospitals. In the case of Noorjahan Hospital, which bought the greatest number of products from these two companies, 1,200 heart disease diagnostic tests conducted at the hospital between November 2015 and June 2016 were compromised by the improperly stored products.
Without the investigation uncovering these problems, hospital employees might never have known about the insufficient cold chain processes. Central Police Hospital, which has a cold chain distribution partnership with VieTech, received medical products in coolers that maintained the proper temperatures. However, based on the investigation, it seems that VieTech was not maintaining those same temperatures prior to delivery to the hospital.
Since uncovering these issues, Bangladesh officials have continued to examine the local medical supply chain. Last month, officials discovered that EM Surgicals was storing antibiotic disks for diagnostic tests at temperatures above the recommended level. In addition, officials confiscated improperly stored blood-clotting drugs, immunosuppressants, human albumin, vasodilators, and hormones used for treating breast cancer and prostate cancer as well as expired drugs and test kits from a variety of distributors.
“This can be called a national disaster,” says Shohael Mahmud Arafat, professor at Bangabandhu Sheikh Mujib Medical University, in the article. “There have been times when a patient has been given insulin but it has no effect on the blood sugar, or administering albumin does not change the protein levels.”
Of course, this is not how cold chain is supposed to work. Islam quotes Anand Shetty, managing director of Novo Nordisk, who explains his company’s cold chain process for transporting and storing insulin. When the drug is imported from Europe, each box is equipped with a thermostat that tracks the temperature of the product throughout the shipping process, he explains. Once the product is received by the company, the temperature logs are checked, and only those products that maintained temperature integrity are approved for further distribution. Any compromised drugs are destroyed. From there, the drug-carrying coolers are sent to distribution centers in authorized cold chain vans that maintain proper product temperatures. Ultimately, the coolers are delivered to pharmacies, where staff is expected to properly store the products until they are needed.
This process breaks down as soon as one player in this supply chain fails to maintain temperature integrity. Unfortunately, this can be hard to track and pinpoint without a robust monitoring system and seamless collaboration among suppliers and customers.
Knowledge is key
Shetty points out that hospitals and pharmacies can uphold their part of the cold chain process by hiring licensed pharmacists who are trained in proper drug storage. The same can be said about manufacturers and distributors employing supply chain professionals who are educated about proper distribution procedures.
According to the APICS Dictionary, distribution includes: “the activities associated with the movement of material, usually finished goods or service parts from the manufacturer to the customer. These activities encompass the functions of transportation, warehousing, inventory control, material handling, order administration, site and location analysis, industrial packaging, data processing, and the communications network necessary for effective management.”
The APICS Certified in Logistics, Transportation and Distribution (CLTD) designation demonstrates an individual’s in-depth knowledge of topics such as capacity planning and demand management, order management, inventory and warehouse management, global logistics considerations, logistics network design, reverse logistics and sustainability, and more. APICS CLTD designees are valuable assets to their companies and help businesses stay competitive in today’s global economy. Start your journey toward certification today at apics.org/cltd.